EV PROCESS DEVELOPMENT AND MANUFACTURING
Extracellular vesicles (EVs) are rapidly emerging as a promising therapeutic platform, capable of delivering a range of APIs including RNA, small molecules and proteins among others. CELONIC now offers reliable and GMP-compliant development as well as manufacturing of extracellular vesicle-based therapeutics and vaccines complete with end-to-end analytics.
EVs are a heterogeneous group of lipid-bound, cell-secreted vesicles involved in intercellular communication. Exosomes and microvesicles, the main subtypes of EVs, are ‘cargo carriers’ as they transport various biomolecules such as proteins, nucleic acids and lipids between cells. Given the immense potential of EVs to address unmet medical needs (e.g. in regenerative medicines and targeted therapies), there is a growing demand for EV development and production under GMP conditions.
OUR EV SERVICES AT A GLANCE
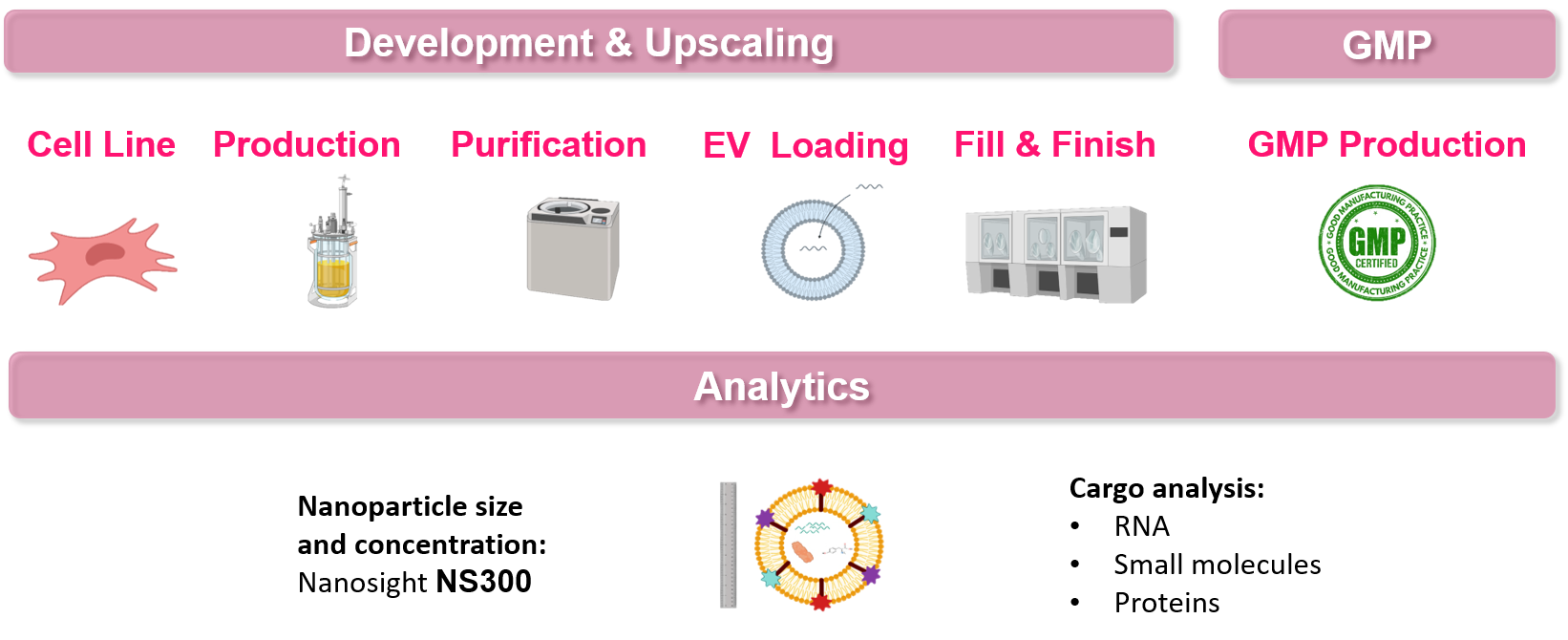
Ev-based biotherapeutic development
CELONIC’s exosome and microvesicle development platforms are fully equipped to transfer, develop and scale-up a broad range of candidate therapeutic as well as vaccine solutions. The capabilities and expertise present at CELONIC provide greater flexibility in project on-boarding, irrespective of development stage. Currently, CELONIC has ongoing EV development projects in both early and late phase development.
CELONIC’s capabilities include:
- Experience with multiple cell lines – native and engineered
- Ability to produce naive, engineered and cargo-loaded EVs
- Established USP and DSP development and scale-up
CELL LINE DEVELOPMENT FOR EV PRODUCTION
CELONIC offers cell line development (CLD) with a wide range of cell lines, including CLIENT-produced cell lines, native and engineered cell lines, as well as the human GEX® cell line – optimized for use in perfusion processing at up to 1000 L scale.
CELONIC’s CLD capabilities include:
- Well-characterized and scalable GEX® platform experience with over forty (40) successful GMP batches
- Regulatory compliance with products expressed in GEX® having been approved by U.S. and European authorities
- High-efficiency as well as high-throughput plasmid transfection
- Automated clone imaging and screening
- GMP MCB generation and maintenance
EV PRODUCTION – USP DEVELOPMENT
CELONIC’s highly experienced upstream process (USP) development team provides robust EV production and scale-up.
CELONIC’s USP capabilities include:
- Suspension and adherent cell culture
- Perfusion and fed-batch cell culture
- Process scaling (up and out)
EV PURIFICATION – DSP DEVELOPMENT
We use state-of-the-art downstream process (DSP) instrumentation and methodologies to achieve EV isolation and purification, including:
- Ultracentrifugation
- Immuno/affinity capture
- Size-exclusion and ion-exchange chromatography
- Tangential flow filtration (TFF)
FLEXIBLE AND TAILORED CLINICAL TO COMMERCIAL GMP MANUFACTURING
CELONIC leverages its more than two decades of experience in GMP manufacturing to offer project-specific solutions to its clients. The significant scientific knowledge and industry experience inherent in our technical and commercial teams ensure a successful client program in strict compliance with international regulatory requirements for emerging therapies.
To ensure expanded manufacturing flexibility, CELONIC offers tailored business models, flexible suite ownership, and rapid facility expansion using modular technologies to meet the unique needs of our clients.
CELONIC offers:
- Multiple molecular modalities with segregated HVAC and unidirectional material, process and personnel flows
- Flexible cleanrooms tailored for individual process scale-up or scale-out requirements
- Pre-clinical to commercial manufacture
EV CARGO LOADING
CELONIC is fully equipped to produce and characterize EVs loaded with a range of cargo molecules including RNA, proteins, and small molecules.
FILL AND FINISH
CELONIC has successfully produced over 300 GMP batches (liquid in a vial) of mAbs, recombinant proteins and vaccines. CELONIC’s Fill and Finish service for liquid-formulated drug products ensures minimal product loss or vial rejection.
CELONIC’s Fill and Finish capabilities include:
- Broad-range of vial sizes and volumes
- Batch sizes from 100 to 3000 vials
- Complete analytical suite for testing and release
- Compliance with EMA and FDA requirements/standards
STATE-OF-THE-ART ATMP PRODUCTION SITES
CELONIC is rapidly expanding its capacity and capabilities for biotherapeutics development and production. In addition to already existing EV facilities in Basel, CELONIC is adding two new state-of-the-art production facilities. One is an expandable, modular production facility for biologics, EVs and mRNA in Heidelberg, Germany and a world-class ATMP production facility in Stein, Switzerland.
END-TO-END ANALYTICS
CELONIC performs comprehensive analytical testing using the latest technologies for EV characterization and quality control,. This includes GMP-compliant method validation and stability studies to enable robust, reproducible and scalable EV assessment and release.
CELONIC’s analytical capabilities include:
- Nanoparticle analysis for determination and characterization of nanoparticle distribution, size and concentration
- Surface marker analysis using FACS, western blot and ELISA
- Cargo analysis incorporating advanced technologies to include qPCR, capillary electrophoresis, UPLC, fluorometry and mass spectrometry
LET’S GET STARTED
We understand what it takes to bring breakthrough EV therapies to patients in need.
Get in touch