Process Development and GMP Manufacturing – Draft GT
Emerging cell and gene therapies require streamlining of viral vector development, production and processing to unlock their full potential. CELONIC now provides expertise and capabilities in process development, scale-up and analytical method development for GMP and non-GMP viral vectors production.
Viral-vector-based gene therapies give patients with inherited single-gene defects, cancers and other previously untreatable diseases new options for safe and effective treatment. A number of cell and gene therapies have successfully moved from bench to bedside, thanks to considerable improvements in efficacy and safety. Scalable, cost-effective and compliant manufacturing will drive continued success of these advanced therapies.
OUR VIRAL VECTOR SERVICES AT A GLANCE
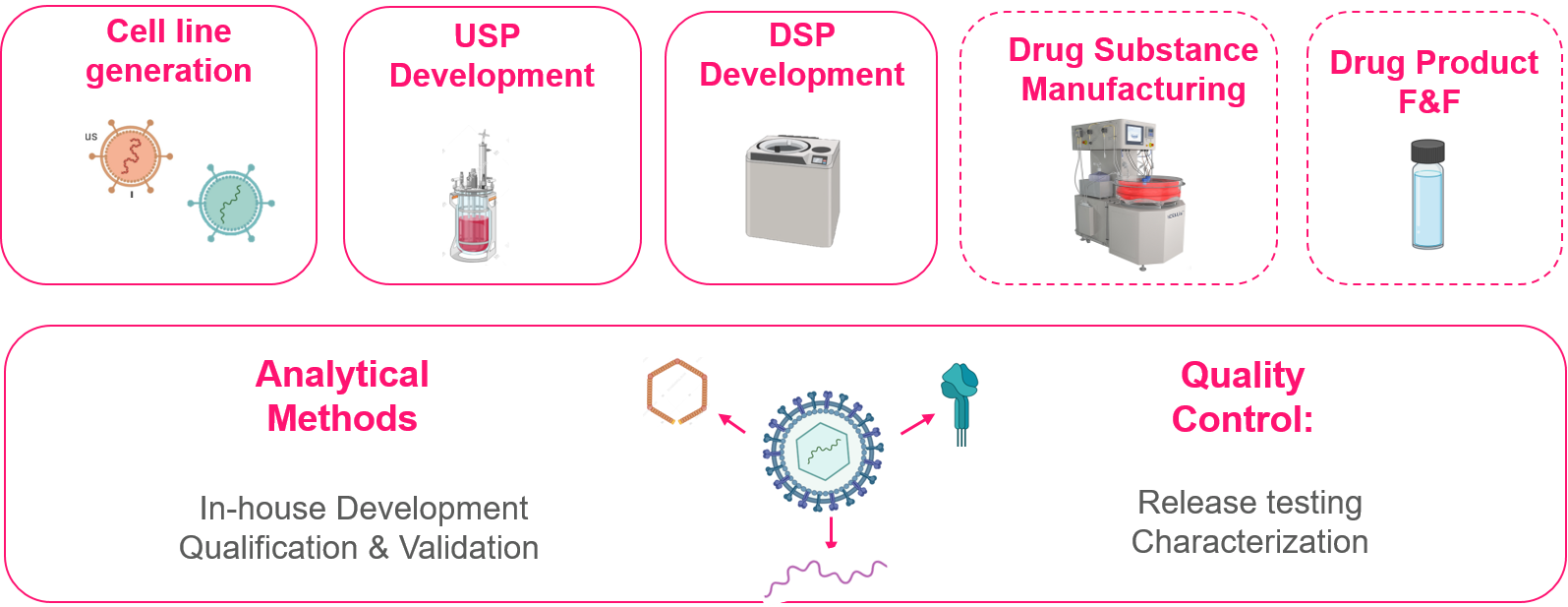
Leveraging its more than two decades of experience in GMP manufacturing, CELONIC supports clients from early phase clinical development through to commercial-scale manufacturing of viral vectors.
Our state-of-the-art viral vector capabilities include:
- Diverse viral vector and cell line platforms
- Multi-scale, high-throughput transient cell line transfection
- Adherent- and suspension-based USP development
- Flexible and project-specific equipment for DSP development
- Comprehensive analytical development
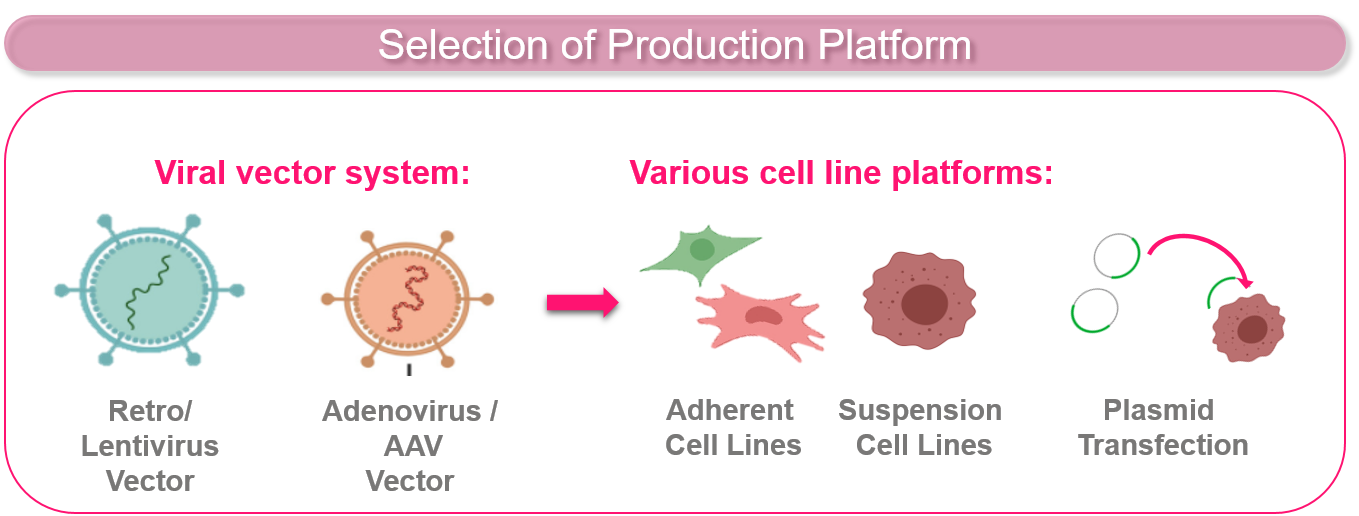
PRODUCTION PLATFORM SELECTION AND CELL LINE TRANSFECTION
CELONIC uses a wide range of viral vector and cell line platforms for adherent- and suspension-based processes.
- Viral vector systems: AAV, Adenovirus, Lentivirus, and Retrovirus
- Cell lines: HEK293, Vero, GEX®
- Transient cell line transfection: inoculation of up to 50 L BR (200 L BR in 2022)
USP AND DSP DEVELOPMENT
CELONIC offers tailored, scalable process development solutions using flexible, project-specific equipment for adherent- and suspension-cell-based processes.
- USP development: equipped to support both small-scale and large-scale GMP processes with scale up to 2000 L BR; iCellis 500 for GMP manufacturing in the new Stein facility in 2022
- DSP development: advanced chromatography and tangential flow filtration equipment for harvest clarification, capture, polishing and concentration.
GMP DRUG SUBSTANCE (DS) AND DRUG PRODUCT (DP) PRODUCTION
Anticipating the unique needs of its CLIENTs, CELONIC offers tailored business models, flexible ownership of suites and rapid modular facility expansion to produce GMP-compliant viral vectors.
- CELONIC’s manufacturing facilities include existing capabilities in Basel, Switzerland and our newest CGT production site with 65,000 ft2 of GMP manufacturing area in Stein, Switzerland
- Compliant fill-finish services for liquid-formulated drug products, including viral vectors
ANALYTICS – VIRAL VECTOR CHARACTERIZATION AND RELEASE
CELONIC provides comprehensive analytical method development using its in-house suite of qualified, leading-edge instrumentation, as well as, a number of qualified external analytical partners.
- Non-GMP in-process control (IPC) – carried out in-house
Assays for identity, purity, titer, and characterization, including mass spectrometry and cell-based potency assays
LET’S GET STARTED
We understand what it takes to bring breakthrough viral vector therapies to patients in need.
Get in touch